Skin Irritation and Corrosion Toxicity Tests: OECD Guideline 439
The safety assessment of products that come into direct contact with the skin is essential for regulatory compliance, particularly concerning human health. Foremost among these assessments are skin irritation and corrosion toxicity tests.
The OECD 439 skin irritation test, developed as an alternative to animal testing, is frequently preferred due to its scientific validity and compliance with legal regulations.
What is OECD 439?
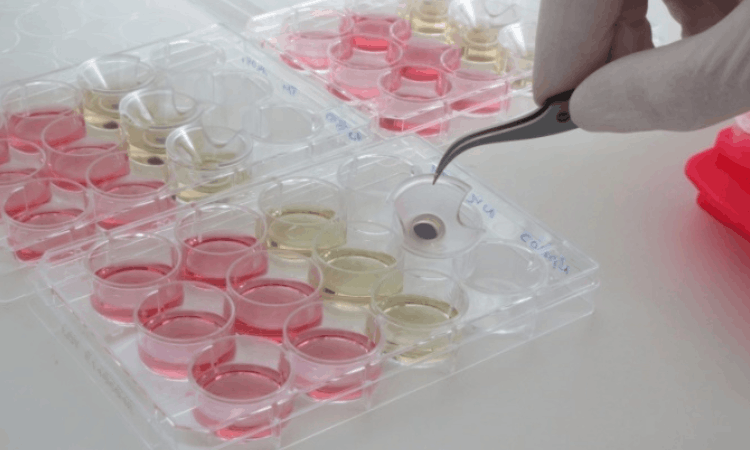
OECD 439 is an in vitro test guideline developed to assess the skin irritation potential of chemicals and products. The test is conducted using Reconstructed human Epidermis (RhE) models, which mimic the human epidermis.
This method offers a reliable alternative, particularly in areas where animal testing is restricted or prohibited.
The Difference Between Skin Irritation and Corrosion
Skin Irritation: Reversible redness, swelling, and irritation of the skin
Skin Corrosion: Irreversible tissue damage penetrating into the deeper layers of the skin
OECD 439 is used to identify irritation, while corrosion assessment is typically supported by methods such as OECD 430, 431, or 435.
Application Steps of the OECD 439 Test Method
- Application of the
- Test substance to the RhE model
- Removal of the sample after the specified contact time
- Measurement of cell viability through metabolic assays
- Evaluation of results based on threshold values
- The determined cell viability rates ascertain whether the product is classified as a skin irritant or not.
For Which Products Is OECD 439 Used?
- Cosmetics and personal care products
- Biocidal Products
- Cleaning and detergents
- Industrial chemicals
- Substances that come into contact with medical devices
- contact
In this regard, OECD 439 forms the basis for safety assessment across numerous industries.
Advantages of the OECD 439 Test
- Alternative to animal testing and ethical approach
- High repeatability
- Short Test Duration
- Cost-Effective Solution
- Internationally Recognized Method
The OECD 439 skin irritation test is a crucial component of modern toxicology, providing a scientific and legally compliant assessment for the protection of human health.
Please contact us to learn about all test details, the requirements for product approval, and the complete procedural workflow.
info@nano-test.de